Theory of Light and Color

32. Absorption, Transmission, and Reflection of Light (Electromagnetic Waves)
As described in Chapters 1–3, light is a type of electromagnetic wave. Electromagnetic waves vary widely in wavelength, from gamma rays with wavelengths less than 1 pm (10⁻¹² m) to radio waves with wavelengths over 1 m. The wavelengths between approximately 10 nm and 1 mm (1 nm = 10⁻⁹ m) are considered "light." Within this range, the wavelengths that cause sensations of "brightness" and "color" in the human eye, roughly 380 to 780 nm, are known as visible light or visible radiation.
Originally, "light" referred only to what could induce sensations of brightness and color in human eyes—essentially visible light. However, as science and technology advanced, we discovered electromagnetic waves just outside the visible spectrum. These waves have similar physical properties to visible light but are invisible to the human eye. As a result, "light" is now understood in a broader sense. ≪1≫
The importance of visible light to human life is undeniable. However, the broader spectrum of light, which includes ultraviolet (UV) and infrared (IR) radiation, also significantly impacts our lives in various ways. These effects—such as skin tanning from UV radiation, the colors of objects from visible radiation, and the thermal effects from IR radiation—differ according to the wavelength of the light. These phenomena may seem unrelated at first glance, but they can all be explained as interactions between electromagnetic waves (light) and the atoms and molecules that make up matter.
(1) Energy Conservation in Reflection, Absorption, and Transmission of Light by Objects
When light (radiant flux) strikes an object, it interacts with the atoms and molecules in the material. This interaction results in the light being divided into three macroscopic outcomes: reflection, absorption, and transmission. The principle of energy conservation applies to these processes.
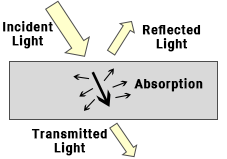
In general, for any object, the incident radiant flux can be expressed as:
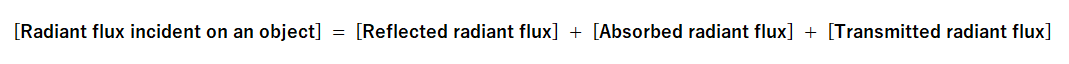
and
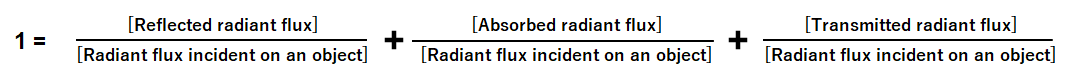
The relative proportions of reflection, absorption, and transmission depend on the material's atomic and molecular composition, the wavelength (frequency) of the incident light, and other conditions such as the angle of incidence and the object surface. For most materials (excluding fluorescent substances), this relationship holds true for each wavelength component of light. Thus, the equation for each wavelength λ is defined as follows:
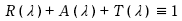
Where:
• R (λ) = spectral reflectance
• A (λ) = spectral absorption
• T (λ) = spectral transmittance
The different colors we perceive in various objects under the same white light are explained by the different spectral reflectance R (λ) and spectral transmittance T (λ) values of those objects in the visible range. (Refer to Chapter 12)
In other words, the color we see is a result of the human eye interpreting the physical phenomena of spectral reflectance R (λ) and spectral transmittance T (λ). These phenomena also occur in the infrared and ultraviolet ranges, not just the visible range. The way light interacts with the molecules and atoms of a material varies depending on the wavelength (frequency) of the light, leading to different observable effects.
(2) Absorption of Electromagnetic Waves (Light) by Matter
One of the key interactions between light and matter is the absorption of light by the material. Light (electromagnetic waves) exhibits both wave-like and particle-like properties. The energy E of a photon (a particle of light) is proportional to its frequency ν and inversely proportional to its wavelength λ. This relationship can be expressed as:
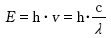
Where:
• h = Planck's constant (h = 6.626×10-34 J·s)
• c = Speed of light in a vacuum (c = 2.998×108 m/s) (c = ν·λ)
Therefore, the energy of a photon increases as its frequency increases (or as its wavelength decreases), leading to the relationship:
Ultraviolet > Visible > Infrared
Atoms and molecules are not stationary; they exist in various energy states due to electronic excitation, molecular vibrations, and rotations. When electromagnetic waves hit a material, and the energy of the incident waves resonates with the energy states of the atoms or molecules, a resonant interaction occurs. This causes the energy of the electromagnetic waves to be absorbed by the material, leading to the activation of the material and the disappearance of the incident waves. This process is known as the absorption of electromagnetic waves by matter.
Three primary energy states of atoms and molecules play a role in this interaction:
[1] Electronic Excitation State: The energy of electrons in an atom exists in discrete levels, known as electronic energy levels. The lowest energy state is called the ground state, and the higher energy states are known as excited states.
[2] Vibrational Excitation State: Molecules vibrate within certain energy levels, including a vibrational ground state and vibrational excited states.
[3] Rotational Excitation State: Molecules can also rotate as a whole while maintaining their structure, with the energy associated with this rotation existing in the rotational ground and excited states.
Each material has its own unique combination of atoms and molecules, leading to specific energy levels for electronic, vibrational, and rotational excitations. When the frequency (or wavelength) of the incident electromagnetic waves matches these energy levels, resonance occurs, and the energy is transferred from the waves to the atoms or molecules, leading to absorption.
These three energy states have different magnitudes, with electronic excitation being the highest and rotational excitation being the lowest. In terms of wavelength:
• Electronic excitation corresponds to energy in the ultraviolet to visible range.
• Vibrational excitation corresponds to energy in the mid-infrared to far-infrared range.
• Rotational excitation corresponds to energy in the microwave or radio wave range, beyond the visible spectrum.
The concept of temperature is related to the degree of vibrational excitation of atoms and molecules. As temperature increases, vibrational activity intensifies. At absolute zero (0 K), atomic vibrations are at their lowest energy state. As energy is added, causing atomic vibrations to increase, the temperature rises. Vibrational and rotational excitations are closely linked to this temperature increase. For instance, infrared heaters convert infrared radiation (from the mid-infrared to far-infrared range) into vibrational energy in the atoms and molecules of objects, causing them to heat up.
Over time, the energy in these excited vibrational and rotational states is released back into the environment as electromagnetic waves, returning the material to its ground state. This release of energy leads to a decrease in the temperature of the material.
Electromagnetic waves in the visible to ultraviolet range, which have shorter wavelengths, have much higher photon energy than required for vibrational excitation and mainly correspond to the energy needed for electronic excitation. When photons in the visible to near-ultraviolet range strike a material, they excite the electrons, resulting in absorption. Since the energy required for electronic excitation varies between materials, the wavelengths of photons that cause this excitation also vary, leading to differences in spectral absorption, and consequently, spectral reflectance and transmittance observed in different materials.
At even shorter wavelengths, in the ultraviolet range, photon energy exceeds that required for electronic excitation, which can lead to the breakdown of molecular bonds within the material. Once these bonds are broken, the material cannot return to its original state, leading to permanent changes. This effect becomes noticeable at the shorter wavelengths of the visible spectrum and becomes increasingly pronounced from near-ultraviolet to far-ultraviolet. For example, prolonged exposure to strong ultraviolet rays from direct sunlight can cause skin inflammation and eye damage, as the near-ultraviolet (UV-A to UV-B) radiation damages the molecular structures in living tissues. Ultraviolet germicidal lamps use even shorter wavelength UV-C radiation to destroy the DNA in bacteria, effectively sterilizing them. However, this radiation is also harmful to human skin and eyes, so it must be handled with care.
By measuring the absorption spectra of different materials, we can analyze and identify them based on their unique electromagnetic absorption characteristics.
(3) Transmission and Reflection of Electromagnetic Waves (Light)
It is often said that objects that appear opaque in the visible light spectrum tend to have higher transmission rates in the near-infrared region. This phenomenon is frequently utilized in machine vision for inspection and analysis. But why does the transmission rate increase in the near-infrared range?
The phenomenon of electromagnetic waves (light) passing through (or being diffusely reflected by) a material is primarily influenced by Rayleigh scattering. Rayleigh scattering occurs when the particle size is much smaller than the wavelength of the incoming radiation, causing the light to scatter (change direction) inversely proportional to the fourth power of the wavelength (or directly proportional to the fourth power of the frequency). Although Rayleigh scattering is commonly used to explain the scattering of light by gases or fine particles in the air (e.g., why the sky is blue, why sunsets are red), it can also apply to solids. ≪2≫
When visible to near-infrared light strikes a material without matching the electronic excitation conditions of its atoms or molecules, absorption does not occur. Instead, the light passes through the spaces between atoms or is scattered. In such cases, infrared light, which has a longer wavelength than visible light, is less likely to be scattered. This means that the direction of the incoming light is less likely to change, resulting in greater transmission through the material. ≪3≫
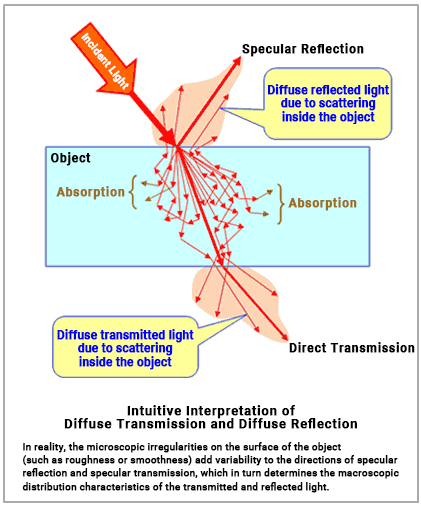
In the case of solids, photons that are not absorbed by the particles on the surface (such as atoms and molecules) of the material enter the interior and interact with other particles within. Short-wavelength photons are more likely to be scattered multiple times, causing their paths to change direction frequently and become more uniform in all directions. In contrast, longer-wavelength photons are less prone to scattering, which means their paths remain more consistent and straight. As a result, in thin materials, more long-wavelength photons can pass straight through (transmission), creating what is known as direct transmitted light. Photons that have their paths altered due to scattering but still exit on the opposite side of the material are seen as diffuse transmitted light. Both direct and diffuse transmitted light together form the observable transmitted light.
Additionally, some photons are reflected directly off the surface of the material in a predictable pattern, known as specular reflection. Besides these, some photons scattered within the material are re-emitted back into the incident space they came from, contributing to the observable reflected light as diffuse reflected light.
Comment
≪1≫
In Japanese, ultraviolet radiation (UV) is called "紫外線" (shigaisen), which means "outside of purple." This refers to wavelengths shorter than the shortest wavelength of visible light (purple). Similarly, infrared radiation (IR) is called "赤外線" (sekigaisen), meaning "outside of red," which refers to wavelengths longer than the longest wavelength of visible light (red). The corresponding English terms are "ultraviolet radiation" (UV) and "infrared radiation" (IR).
However, when we look at the meanings of the prefixes "ultra-" and "infra-" in English, it can seem a bit confusing. The prefix "ultra-" means "beyond" or "above," while "infra-" means "below" or "within." Based on these meanings, one might expect that ultraviolet and infrared would be reversed in terms of wavelength.
This confusion comes from the fact that today, we often talk about light in terms of its wavelength. However, understanding the historical context of how broad-spectrum light (including ultraviolet and infrared radiation) was discovered and studied helps explain why these terms were chosen.
Ultraviolet and infrared radiation were discovered through experiments on how sunlight and other forms of radiation affect different substances. These studies revealed that there are types of radiation outside the visible spectrum that are invisible to the human eye. Early research focused on the energy of light, which is related to its frequency, and how it affects materials. Ultraviolet radiation has higher photon energy than visible light, so it was called "ultraviolet" (beyond violet). Infrared radiation has lower photon energy, so it was named "infrared" (below red). This naming makes sense when we consider that the effects of light on objects depend more on the energy carried by the light than on its wavelength. (It's also worth noting that, from the time of Newton until around the 18th century, light was often considered as particles.)
≪2≫
The density of particles that cause scattering in solids is significantly different from that in gases. Whether a solid is metallic or non-metallic (and whether it contains free electrons) also influences how scattering behaves. Therefore, it may be overly simplistic to explain scattering in solids using only the rule that scattering is inversely proportional to the fourth power of the wavelength. However, it is generally accepted that similar scattering phenomena still occur.
≪3≫
In the mid-infrared and far-infrared regions, absorption tends to increase because these wavelengths correspond to the vibrational and rotational excitation bands of molecules. This increase in absorption leads to a decrease in transmittance in these regions.

